|
6/15/2023 0 Comments Southern blot analysis
Suppose, for example, that the c- abl probe ordinarily recognizes a 2,000-base EcoRI fragment in normal genomic DNA. Therefore, if a gene has undergone a structural rearrangement, as, for example, when the c- abl oncogene is translocated from chromosome 9 to chromosome 22, the pattern may change. For any particular gene probe, the size (ie, length) of the band it identifies will be the same from individual to individual (although see below for a discussion of restriction fragment length polymorphism, an important exception). Each band corresponds to a restriction endonuclease-generated DNA fragment containing nucleotide sequences complementary to those in the radioactive probe. What results from these studies is a pattern of one or more bands on radiographic film. The unbound probe can be washed away, and the remaining specifically hybridized probe can be visualized by exposing the filter to radiographic film. If there are any sequences in the genomic DNA that are complementary to those in the probe, the probe will hybridize to those sequences on the filter. The membrane containing the transferred DNA is then soaked in a solution containing the radiolabeled probe. After transfer, the DNA fragments are arrayed by size on the solid support.Īt this point, a fragment of cloned DNA (the probe) is radiolabeled by using any of a variety of techniques. As the liquid perfuses the gel, it carries DNA fragments with it, depositing them on the membrane filter, to which the DNA sticks. Liquid is then forced through the agarose gel in a direction perpendicular to the direction in which the DNA moved during electrophoresis. Thus, after electrophoresis, a paper-thin membrane microfilter (made of nitrocellulose or nylon) is placed over the flat portion of the gel. The DNA fragments must be transferred to a solid support to which they are irreversibly bound in order to carry out nucleic acid hybridization studies. Because the agarose gel used in electrophoresis is thick and the DNA fragments can move within it, DNA in the gel is not in a suitable form for further analysis. The final goal of Southern blotting is to identify specific fragments of cut DNA by using nucleic acid hybridization. Digested DNA is arrayed by size using electrophoresis through a semisolid (more.) Genomic DNA is digested with a single restriction endonuclease resulting in a complex mixture of DNA fragments of different sizes, that is, molecular weights. At any time after electrophoresis begins, small molecules will be closer to the anode than will large molecules.) The agarose gel is usually cast in the form of a flat rectangle a few millimeters thick. The semiporous agarose allows molecules of DNA to pass with varying degrees of ease, at a rate inversely proportional to their size. 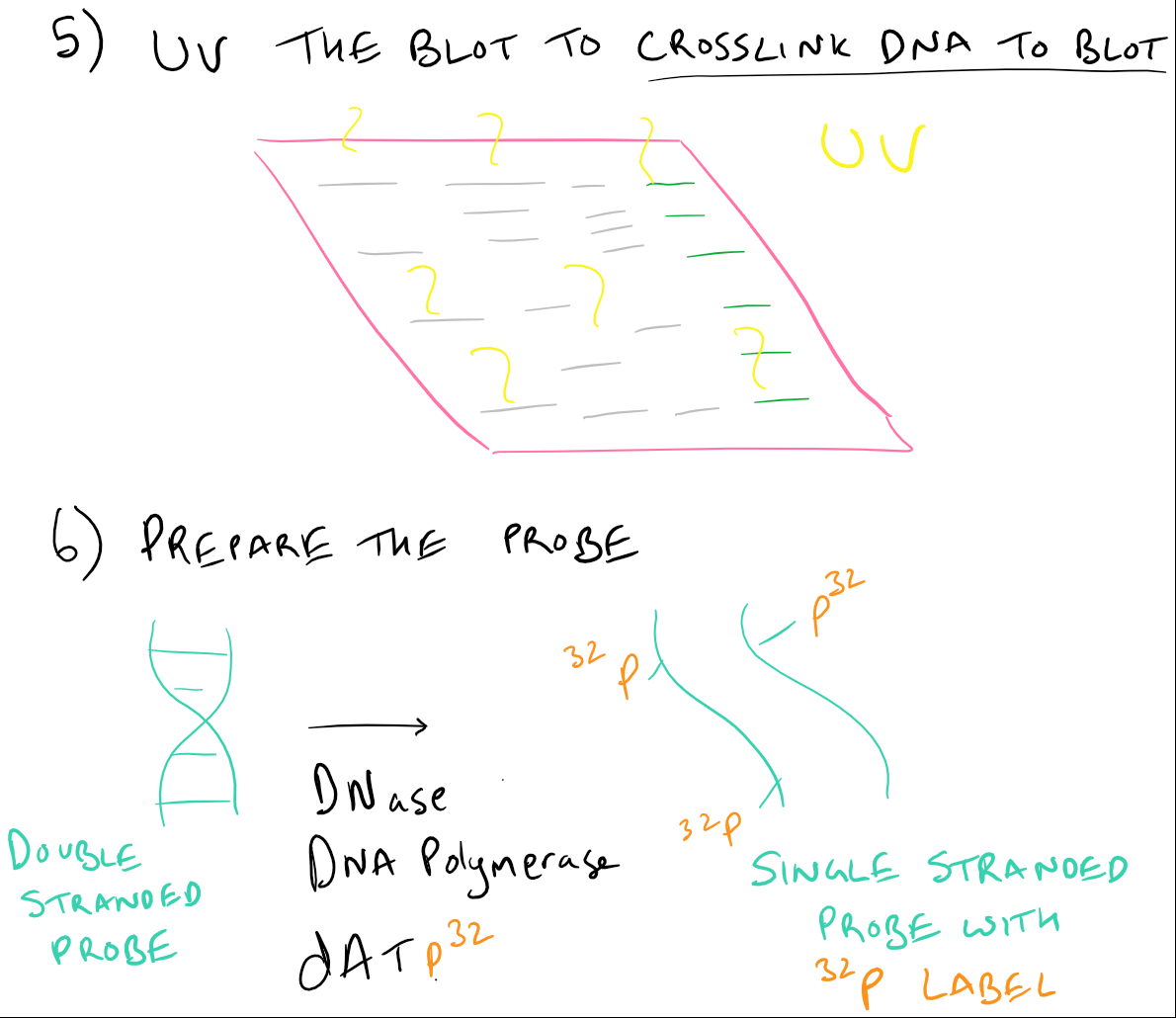
(Because the phosphate groups in DNA make the molecules negatively charged, they will migrate toward the anode in an electric field. Electrophoresis through an agarose gel then separates these fragments according to size. Purified genomic DNA is digested with a specific restriction endonuclease, which, as described above, will produce an array of differently sized DNA fragments. The presence of these sequences usually means that the gene itself is present in the genomic DNA. 17 In general, it allows one to determine whether specific nucleotide sequences in a cloned probe are present in a sample of genomic DNA. One of the most useful techniques for analyzing a gene at the level of genomic DNA is Southern blotting, named for its originator, E.M.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
